OPTIMIZATION OF THE SYNTHESIS OF 2-(2-HYDROXYPHENYL)BENZAZOLE DYES IN POLYPHOSPHORIC ACID
Keywords:
carbon–nitrogen bond formation, cyclodehydration, nitrogen heterocycles, ESIPT dyesAbstract
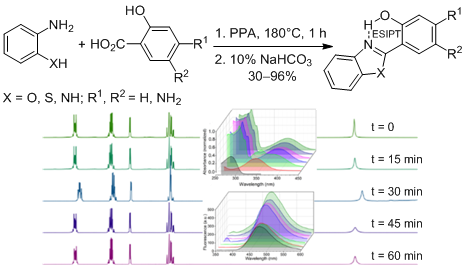
2-(2-Hydroxyphenyl)benzazole dyes are particularly interesting molecules that exhibit excited-state intramolecular proton transfer (ESIPT) following photoexcitation. In 1957, Hein employed polyphosphoric acid as both a solvent and dehydrating agent to synthesize a series of 2-aryl-substituted benzimidazoles, benzoxazoles, and benzothiazoles, which had previously been inaccessible. However, the harsh reaction conditions for an extended period produce side reactions, and even carbonization inevitably can occur. Since this method continues to be used widely, in our work we have done time-dependent experiments to verify whether it would be possible to shorten reaction time. For this, the synthesis of a series of 2-(2-hydroxyphenyl)benzazole dyes from anilines ortho-substituted with aromatic carboxylic acids in polyphosphoric acid was monitored over time through 1H, 13C NMR, UV-vis, and fluorescence spectroscopies. Our results showed that all reagents were consumed, and the expected dyes were formed within an hour of reaction with yields ranging from 30 to 96%. It was demonstrated that the shorten reaction time results in fewer thermal decomposition products. Furthermore, the reduced reaction time does not significantly affect the yields, and no additional purification procedure is required.