EFFICIENT SYNTHESIS OF 3-BORYLATED INDOLINES VIA COBALT-CATALYZED DEAROMATIVE BORYLATION OF INDOLES
Keywords:
borylation, dearomatization, indole, indoline, cobalt(II), terpyridine, trifluoroboratesAbstract
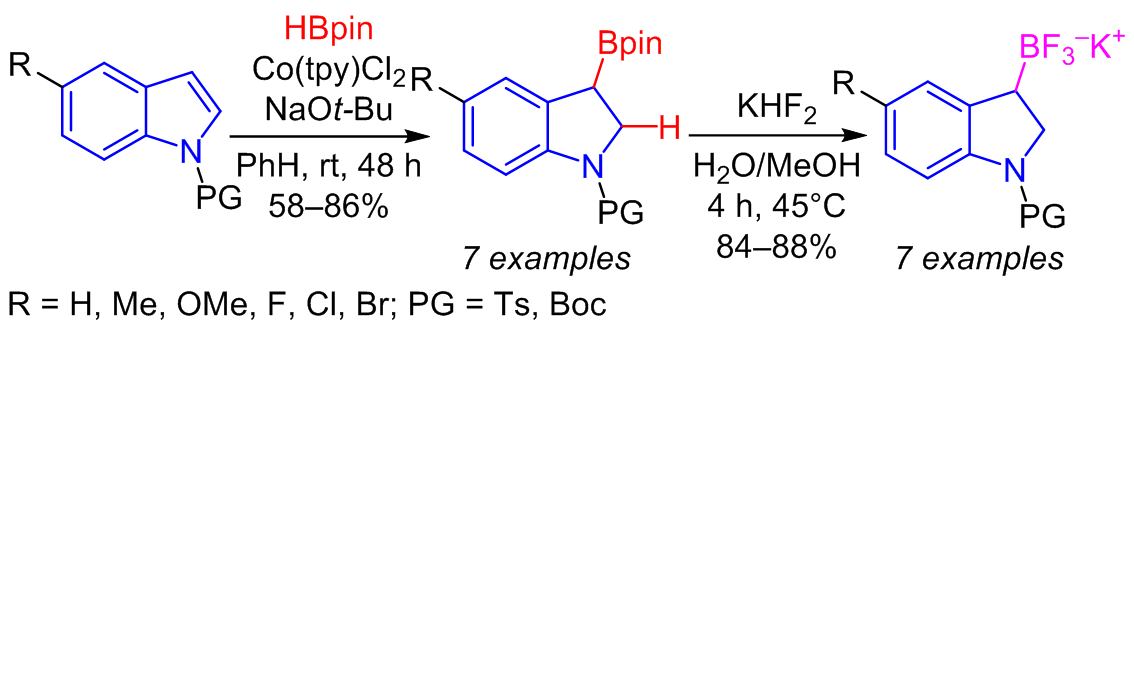
The high efficiency of the cobalt(II) terpyridine complex as a precatalyst in the dearomative borylation of indoles using pinacolborane as the borylation agent was demonstrated. The reaction was tested on a series of indoles bearing substituents of different nature at the C-5 position of the indole ring and with various electron-withdrawing protecting groups on the indole nitrogen. The applicability of the proposed synthetic protocol for gram-scale synthesis was also demonstrated. The resulting pinacolboranes were subsequently converted into the corresponding boron fluorides with high yields.
Downloads
Additional Files
Published
2026-02-24
Issue
Section
Original Papers
