5-CHLORO-4-[(THIAZOLIDIN-5-YLIDENE)METHYL]PYRROLE-3-CARBOXYLATES AND CARBOXYLIC ACIDS: SYNTHESIS, RECYCLING, AND ANTIMICROBIAL ACTIVITY
Keywords:
5-chloro-4-formylpyrrole-3-carboxylates, 5-chloro-4-formylpyrrole-3-carboxylic acids, 5-chloro-4-[(thiazolidin-5-ylidene)methyl]- pyrrole-3-carboxylates, 5-chloro-4-[(thiazolidin-5-ylidene)methyl]pyrrole-3-carboxylic acids, thieno[2,3-b]pyrrole-2,4-dicarboxylic acids, alkaline hydrolysis, antimicrobial activity, recyclingAbstract
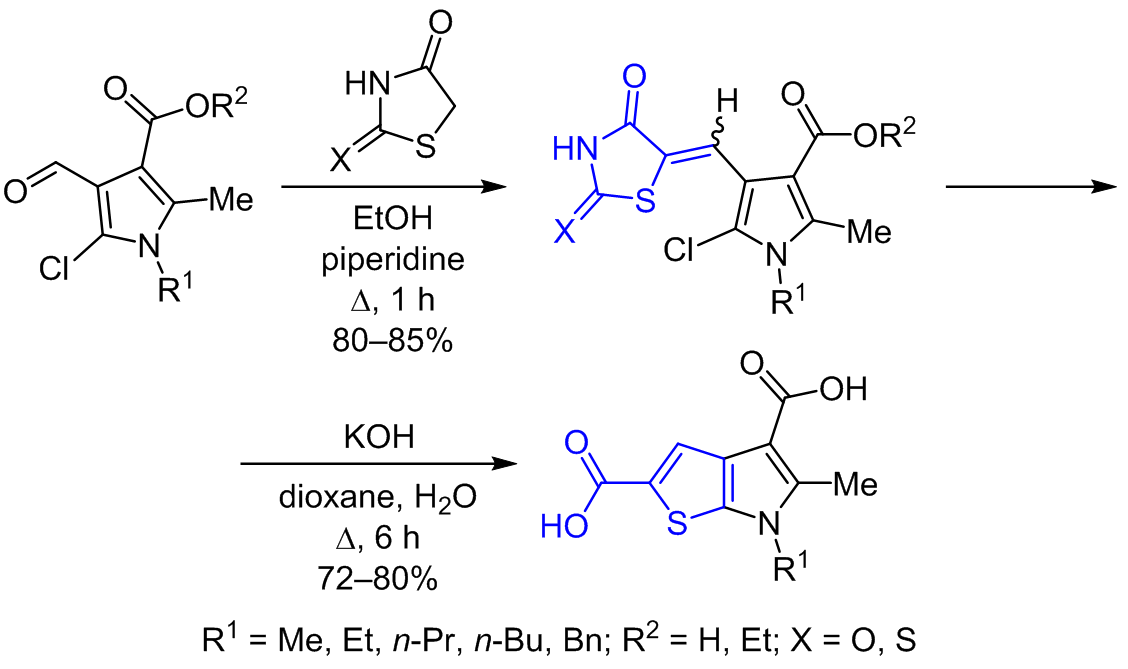
5-Chloro-4-formylpyrrole-3-carboxylates and carboxylic acids can smoothly interact with thiazolidin-4-ones and form 5-chloro-4-[(thiazolidin-5-ylidene)methyl]pyrrole-3-carboxylates and carboxylic acids, respectively, which can further be transformed into thieno[2,3-b]pyrrole-2,4-dicarboxylic acids through the alkaline hydrolysis. Moderate antimicrobial activity has been registered in 5-chloro-4-[(thiazolidin-5-ylidene)methyl]pyrrole-3-carboxylates and the corresponding carboxylic acids.
Downloads
Additional Files
Published
2026-02-24
Issue
Section
Original Papers
