RATIONAL DESIGN, SYNTHESIS, AND CATALYTIC ENANTIOSELECTIVE DERIVATIZATION OF AN ACHIRAL ANSA-MACROCYCLE
Ключевые слова:
asymmetric catalysis, atropisomer, design and synthesis, macrocycle, planar chirality, pyrrole-based compoundАннотация
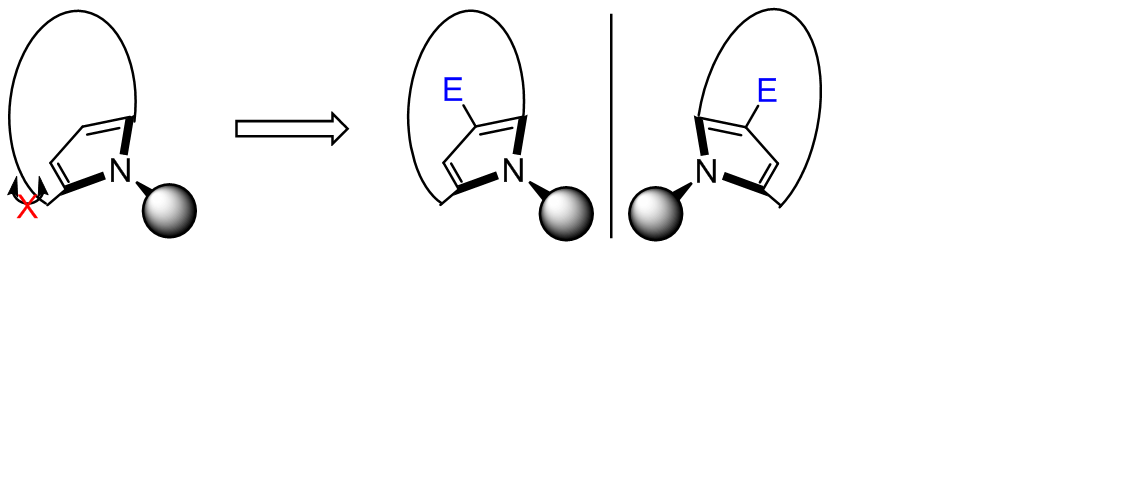
A pyrrole-based achiral atropisomeric macrocycle was rationally designed. Density functional theory calculations predicted that it possesses atropisomeric rigidity comparable to common cyclophanes, while exhibiting enhanced nucleophilicity. This combination indicates broad application potential in planar chiral derivatization. Achiral macrocycle has been successfully synthesized, and nuclear magnetic resonance studies confirmed its atropisomeric nature. Furthermore, the planar chiral derivatization of achiral macrocycle with electrophiles has been investigated. Under the chiral phosphoric acid catalysis, this derivatization proceeded with moderate stereoselectivity.
Загрузки
Дополнительные файлы
Опубликован
2026-02-24
Выпуск
Раздел
Краткие сообщения
