THE ROTATIONAL BARRIER AND THE SUBSTITUENT EFFECT ON RELATIVE STABILITY OF MONOSUBSTITUTED PYRIDINES AND PYRIMIDINES
Ключевые слова:
pyridine, pyrimidine, noncovalent interactions, rotational barrier, stabilization energy, substituent effectАннотация
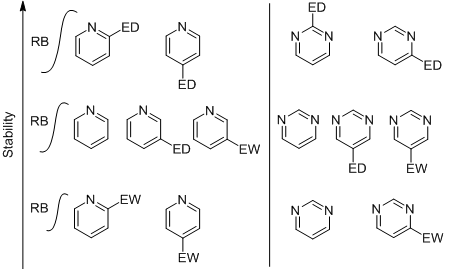
This theoretical study investigates the relative stability of various isomers of monosubstituted pyridines and pyrimidines, focusing on those modified by strong electron-withdrawing and electron-donating groups. The research centers on measuring the rotational barrier associated with the bond linking the substituent to the pyridine or pyrimidine ring, as this parameter is critical for assessing both the stability and the degree of electron delocalization within these compounds. The positional stabilities of the isomers are analyzed by correlating rotational barrier values with a range of structural, atomic, and spectroscopic characteristics, alongside stabilization energy and noncovalent interactions. Notably, electron-donating groups located in the positions 2 and 4 exhibit relatively high rotational barrier values, promoting electron delocalization through resonance and inductive effects. In contrast, electron-withdrawing groups tend to lower rotational barrier values, leading to destabilization of the pyridine and pyrimidine rings, particularly when positioned ortho or para. This study can be extended to 5-membered heterocyclic compounds with different heteroatoms, as well as to multisubstituted heterocyclic systems.