NITRILES AS MULTIPURPOSE REAGENTS FOR THE SYNTHESIS OF SULTAMS AND SULTONES
Ключевые слова:
sultams, nitriles, cyano group, cyclization, sulfonamidesАннотация
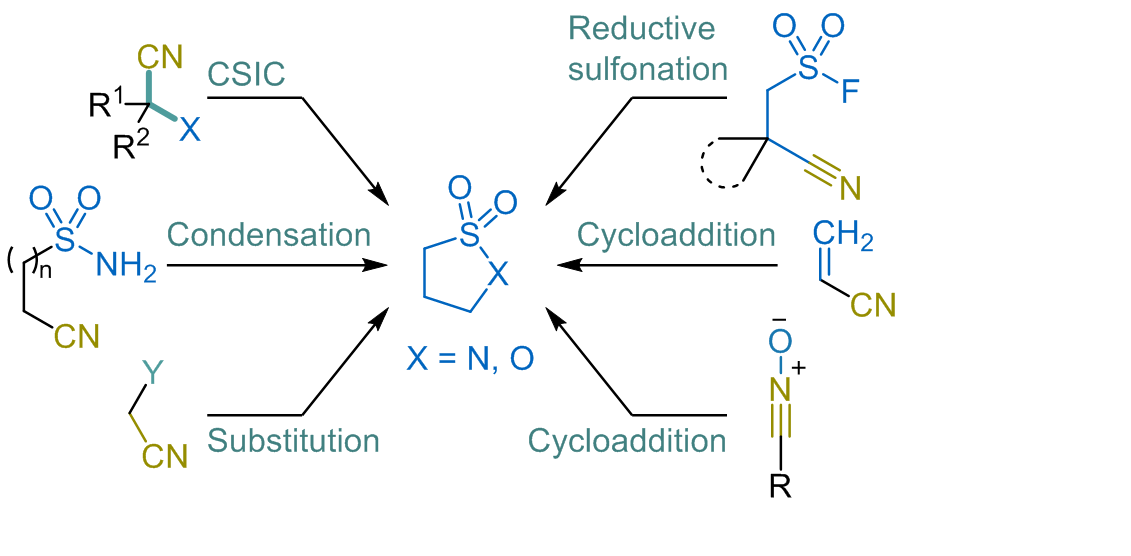
In this review, we discuss the use of the nitrile function in the synthesis of cyclic sulfonamides (sultams), an important class of antibiotics. The cyano group is a versatile building block in sultam synthesis. It can form the sultam and sulton skeleton or activaten adjacent positions. Commonly, the nitrile group undergoes the carbanion-mediated sulfonate (sulfonamide) intramolecular cyclization reaction, acting as an electrophilic center attacked by a carbanion, forming enamines. Nitriles also facilitate substitutions at α-position through alkylation or cyclization and activate unsaturated bonds in various cycloadditions. Literature indicates that the nitrile group, as a methyleneamine precursor, shows promise for sultam synthesis. This method could efficiently produce spirocyclic sultams with applications in chemistry and biology.