A SYNTHESIS OF POLYFUNCTIONALLY SUBSTITUTED CARBO- AND HETEROCYCLIC COMPOUNDS BASED ON A REACTION OF ETHYL ARYLMETHYLIDENECYANOACETATE WITH ACETOACETIC ACID N-ARYLAMIDES
Ключевые слова:
арилметилиденциануксусный эфир, N-ариламиды ацетоуксусной кислоты, аддукт Михаэля, ретро-реакция Михаэля, внутримолекулярная циклизацияАннотация
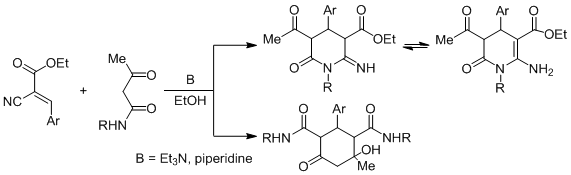
The reaction of ethyl arylmethylidenecyanoacetate with acetoacetic acid N-arylamides was studied. It was revealed that in the presence of catalysts (triethylamine, piperidine) in ethanol (acetonitrile), the dominant direction of the reaction was the formation of the products of the retro-Michael reaction, new oxocyclohexane-1,3-dicarboxamide derivatives. It was established that, along with carbocyclization, an intramolecular heterocyclization of the Michael adduct at the nitrile group was also realized with the formation of previously unknown ethyl 5-acetyl-1,4-diaryl-2-imino-6-oxo-piperidine-3-carboxylates and ethyl 5-acetyl-2-amino-1,4-diaryl-6-oxo-1,4,5,6-tetrahydropyridine-3-carboxylates. The antibacterial activity of some of the obtained compounds was investigated.