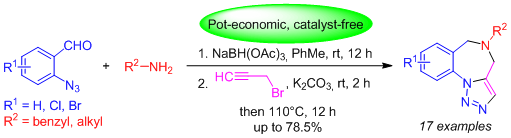
ONE-POT REDUCTIVE AMINATION OF 2-AZIDOBENZALDEHYDES WITH ALIPHATIC AMINES, N-PROPARGYLATION AND INTRAMOLECULAR (3+2) CYCLOADDITION FOR THE SYNTHESIS OF N-SUBSTITUTED TRIAZOLOBENZODIAZEPINES
Ключевые слова:
triazolobenzodiazepine, (3+2) cycloaddition, reductive amination, synthesisАннотация
An efficient and pot-economic synthetic approach toward N-substituted triazolobenzodiazepines is presented. This efficient one-pot synthesis, involving the reductive amination of 2-azidobenzaldehydes with benzyl/alkyl amines, followed by N-propargylation and intramolecular (3+2) cycloaddition, generates a variety of N-substituted triazolobenzodiazepines in satisfactory yields.